Q&A: Bruce Gates on the Molecules That Can Drive Chemical Reactions
The professor at University of California, Davis, describes his innovative work at SLAC’s synchrotron to search for simple, selective catalysts.
By Amanda Solliday
There’s a class of materials responsible for the chemistry we rely on to make fertilizer for crops, create prescription drugs and refine oil into gasoline. They’re called catalysts, and they speed up chemical reactions and steer the direction of the changes that happen during the transformation from one chemical compound to another. Despite the fact that many catalysts are commonly found in biology (these catalysts are called enzymes), the chemistries of most catalysts are still not fully understood because of their complexity.
Bruce Gates, a professor of chemical engineering at the University of California, Davis, specializes in this class of materials.
We asked him about his most recent work at the Department of Energy’s SLAC National Acceleratory Laboratory, where he’s teamed up with researchers at the Stanford Synchrotron Radiation Lightsource (SSRL) to develop new approaches that take a close look at the fine details of these materials and how they perform.
Q: What makes a good catalyst?
There are several criteria for how good a catalyst is. Selectivity is an important goal—make more of the desired substance and less of the undesired substance during chemical reactions.
Another criterion for a catalyst is activity. The more active a catalyst is, the faster it makes a chemical reaction happen. You can get more product over a period of time if the catalyst is highly active.
Another is stability. That’s the issue of how long a catalyst lasts. They don’t last forever, but if they could be made to last substantially longer, while being active and selective, then there’s an economic advantage.
Chemical reactions happen on the surface of a catalyst. In the end, we’re striving for a deep fundamental understanding of the structure of the active site on the catalyst surface and how it influences activity, selectivity and stability. And if we can succeed with that, guided by theory, then we have a platform for making good predictions about how well a catalyst can accelerate a reaction we are interested in.
Q: What else do consider when you create or examine a catalyst?
With any technology, we have to be thinking about cost, environmental concerns, and of course safety.
It may be that the catalyst that is good in terms of those primary criteria happens to be very expensive. The catalytic converter in your car contains platinum, for example. (People steal catalytic converters because of the value of the platinum.) If it happens to be cheaper, it may be a better catalyst even though it’s not so active, selective and stable.
Another concern that’s always important has to do with environmental issues. A catalyst may have toxic components. After the catalyst comes to the end of its useful life, how do we dispose of it in a proper way? That challenge might be so big and expensive, that the catalyst may not actually be useful.
And regarding safety, some catalysts used in industry are pyrophoric – if they’re exposed to air, they will burn. They need to be handled properly to work safely as catalysts.
Q: How are catalysts studied?
It’s been a challenge – that’s a bit of an understatement – to well understand what catalysts are and how they work.
One important research approach is to make catalysts through very careful synthesis that have a structural simplicity or a structural uniformity. That means that we can try to understand a simple material in some depth, because it is easier to study than a more complicated material.
There’s been a lot of wonderful work done in years past investigating simple catalysts that people can understand well. These catalysts were single crystals of metal, for the most part, and Gerhard Ertl won a Nobel Prize in 2007 for this type of work.
But what we do is different. We want to look at how actual catalysts work under realistic industrial conditions.
Q: How is your research group looking at catalysts?
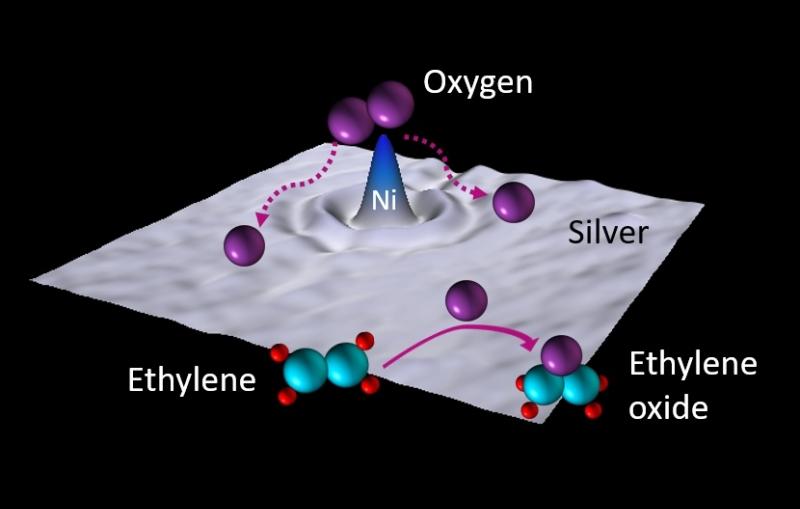
Our approach is to see if we can make a solid catalyst as simple as possible. To do this, we create conditions where the part of the catalyst molecule containing a metal atom of known structure reacts with the surface of a solid, which we call a support.
If we choose the support well, that means we need it to be highly uniform to present a set of sites on its surface that in the best of cases are all the same. We can approach that ideal by using solids that are crystalline, porous materials. We’ve had the most success with zeolites, which are porous minerals made of aluminum, silicon and oxygen. Zeolites are very important catalysts in their own right, and hugely important in petroleum refining and petrochemical conversion. And for us, they’re also important as supports for the catalysts we create.
What we’re striving for is fundamental understanding. And what we’re seeing on the horizon is a class of catalysts that have unique sites on their surfaces and will offer the prospects of high selectivity. And there are opportunities to make more practical catalysts of that class.
Q: Why do you need a synchrotron to do this work?
For years, we’ve done experiments at SSRL that look at catalysts like the ones I’ve described, and watch these catalytic reactions in real time under practical conditions. Our goal is to know what is in those samples, exactly, and how the materials behave as catalysts while they are actively working.
For instance, we’ll put the material we created in a small compartment located in a beam line of the synchrotron, and then we can make that system a “flow-reactor”. Gas molecules that will change during the course of the reaction flow through the system and come in contact with the support and the metal atoms bonded onto its surface. We can see how the catalyst is functioning, that is, how fast it converts some molecules to other molecules as the reaction happens, and at the same time learn what its structure is.
A collaboration between our research team and SSRL developed the use of a special technique with a long name – high-energy resolution fluorescence detection (HERFD) X-ray absorption spectroscopy – to get very fine-grained information about the structure of the catalysts. It’s providing a new kind of data that is especially informative for those types of catalysts that I’ve described—one that’s structurally highly uniform, and has well-defined sites on its surface.
Q: What’s next for your research team?
There’s substantial effort to upgrade and improve the research on catalysis that’s done at SSRL, with leadership by Simon Bare. Simon is distinguished staff scientist at SSRL and an adjunct professor in the Department of Chemical Engineering at the University of California, Davis.
We are looking forward to lots of good things emerging from the growth and improved capabilities of SSRL that make the facility valuable to people in the catalysis community. There’s a lot of opportunity to do some wonderful catalysis science at a synchrotron. It’s on the upswing.
SSRL is a DOE Office of Science user facility.
For questions or comments, contact the SLAC Office of Communications at communications@slac.stanford.edu.
SLAC is a multi-program laboratory exploring frontier questions in photon science, astrophysics, particle physics and accelerator research. Located in Menlo Park, Calif., SLAC is operated by Stanford University for the U.S. Department of Energy's Office of Science.
SLAC National Accelerator Laboratory is supported by the Office of Science of the U.S. Department of Energy. The Office of Science is the single largest supporter of basic research in the physical sciences in the United States, and is working to address some of the most pressing challenges of our time. For more information, please visit science.energy.gov.